The Complicated Link Between Rheumatoid Arthritis and Lung Disorders
What is Bronchiectasis (BR) and how it is related to Rheumatoid Arthritis (RA)? It is known that there is a strong association with RA and various pulmonary disorders such as BR. How these two conditions are connected is less clear and something that has been being examined for some time. However new findings are constantly being revealed. Recently researchers have identified antibodies in BR patients that could help predict whether or not they will develop RA.
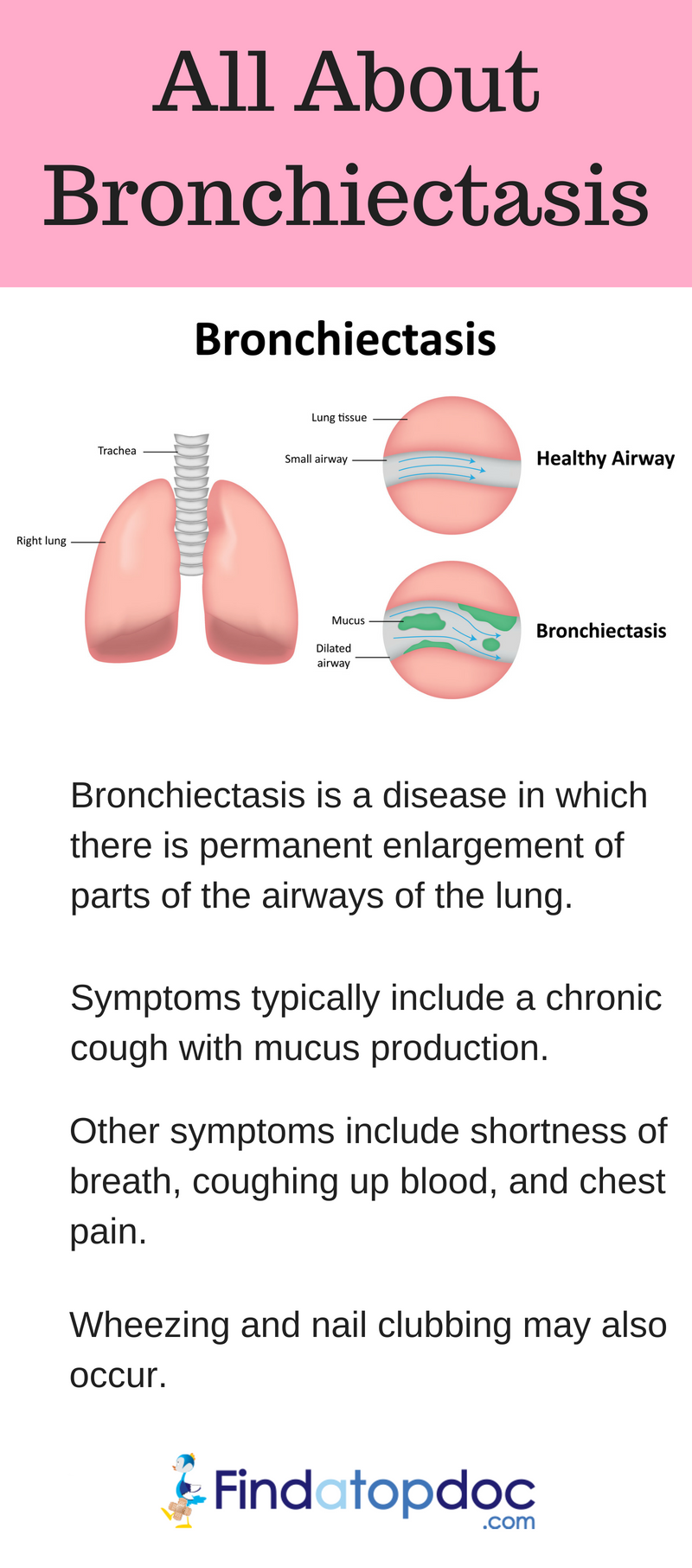
What is Bronchiectasis?
BR is not a common lung condition and it usually develops from a previous infection. It causes damage to the inner structures of the lungs called bronchi. It often causes a cough that can last for months to years with the possibility of coughing up blood and patients experiencing additional symptoms such as pain, fever, and more. It is usually diagnosed through a thorough patient history, chest X-ray, and an analysis of chest sputum. Treatment often involves antibiotics, corticosteroids, bronchodilators and much more depending on the patient and the severity. Patients can do well if they are compliant with treatment plans and work to prevent infections and additional lung conditions. It is chronic condition that requires constant management and evaluation of the patient’s health care plan.
What is the relationship to RA and what’s the path forward?
A literature review published in 2013 evaluates the relationship between RA and BR. The authors acknowledge various limitations to their study at the beginning of the publication. These include variations in diagnostic criteria among the studies that they reviewed as well as different patient profiles including “age, sex, use of nonbiological and biological disease modifying anti-rheumatic drugs (DMARDs), duration of RA and BR, underlying pulmonary disease, and smoking status.” These limitations are important to note because they can affect the validity of the study. The authors explain that the presence of BR in the population of people with RA seems to be at about two to three percent, but various studies report different statistics.
While the relationship between RA and BR has been known for a while, scientists and medical professionals have not fully understood the etiology of this relationship. There are many different theories that have been looked at. One notable finding that this study reports is that RA seems to usually come before BR, and subsequent development of BR does not appear to influence the severity of the RA. One study that was analyzed in this review revealed that subjects in the antibody positive groups had significantly worse pulmonary symptoms than the control groups. This means that the people with autoimmunity in RA had worse pulmonary complications than those with negative antibodies or the control group. The authors concluded that autoimmune related injury may initially start in the lungs. This literature review echoes the information brought forward in the recent article by Joana Fernandes PhD.
Lack of treatment and care research
The literature review also emphasizes that there is a lack of information regarding how to best manage and care for patients with both RA and BR. At the present moment they recommend that patients receive care from specialists for both conditions and that current guidelines be followed. They also discuss the importance of patients on DMARDs and corticosteroids for RA being monitored closely as these drugs can increase the risk for infection and thus possibly development of BR.
In summary the authors concluded that BR has been found to be present on average about 16.4-28.6 years prior to RA development. Additionally, the onset of RA in BR patients was much earlier than onset in people without BR. While they did not form a conclusion on the exact details of how the two conditions are related, they recognize that it likely involves in some way an immunological process and perhaps even a genetic predisposition. Ultimately further research is needed.
Joana Fernandes PhD has recently highlighted additional aspects of the relationship between BR and RA. She writes that the levels of some antibodies that the body produces against two proteins may offer insight into predicting the risk of RA development in BR patients. These two proteins are called calreticulin (CRT) and citrullinated calreticulin (citCRT). The study that she reviewed is “Heightened autoantibody immune response to citrullinated calreticulin in bronchiectasis: Implications for rheumatoid arthritis.” The inflammation that is a hallmark of RA is triggered by CT and citCRT. These proteins do this by binding to other molecules in cells. The authors of the study that she reviewed said that “recently we have demonstrated that RA patients with BR have increased disease activity, severity and autoantibody positivity and the RA autoantibodies are predictors of RA developing in BR patients.” This highlights a change in evidence compared with the 2013 literature review where authors said that there was not yet evidence to support that development of BR does not exacerbate RA symptoms.
The authors studied both RA patients with and without BR to ensure that a control group was present. This enabled them to better understand the level of citCRT in individuals and the resulting antibody response to CRT and citCRT. They also looked at additional risk factors that patients may have had such as smoking and lung disease.
The researchers looked at the levels of the antibodies produced by the body against CRT and citCRT in a population of 388 subjects. About one eighth of the subjects were RA patients, and a little over one fourth were BR patients. One fourth of the subjects were BR patients with RA and there were also 87 asthma patients and 77 healthy individuals in the study. The results showed that “eighteen percent of BR patients with or without RA had CRT antibodies in their blood, and thirty-five percent of BR or RA patients had citCRT antibodies compared to fifty-eight percent of patients with both diseases.”
What do the results mean for the future?
During the next four months the subjects were followed closely. Of note, seventy-five percent of BR patients who developed RA during this time had citCRT antibodies in their blood. These results are important because they reveal a possible biomarker that could be used to predict RA development in BR patients. This could aid providers in assessing the risk of their BR patients developing RA. Ultimately more research is needed to establish the accuracy of these potential biomarkers. What is also of interest, is that this could be paving the way for potential new treatments that target these proteins.